Every vaccine tells the same fundamental lie: it convinces your immune system that a dangerous pathogen has arrived, without the danger. What happens next is one of the most sophisticated biological processes in the human body, a multi-stage training exercise that leaves your defenses permanently upgraded against a threat they may never have truly encountered.
Understanding how this works matters beyond academic curiosity. In an era of mRNA vaccines, pandemic preparedness debates, and growing questions about booster schedules, the molecular machinery behind acquired immunity is no longer just immunology. It is public infrastructure.
The First Alarm: How Your Body Detects a Vaccine
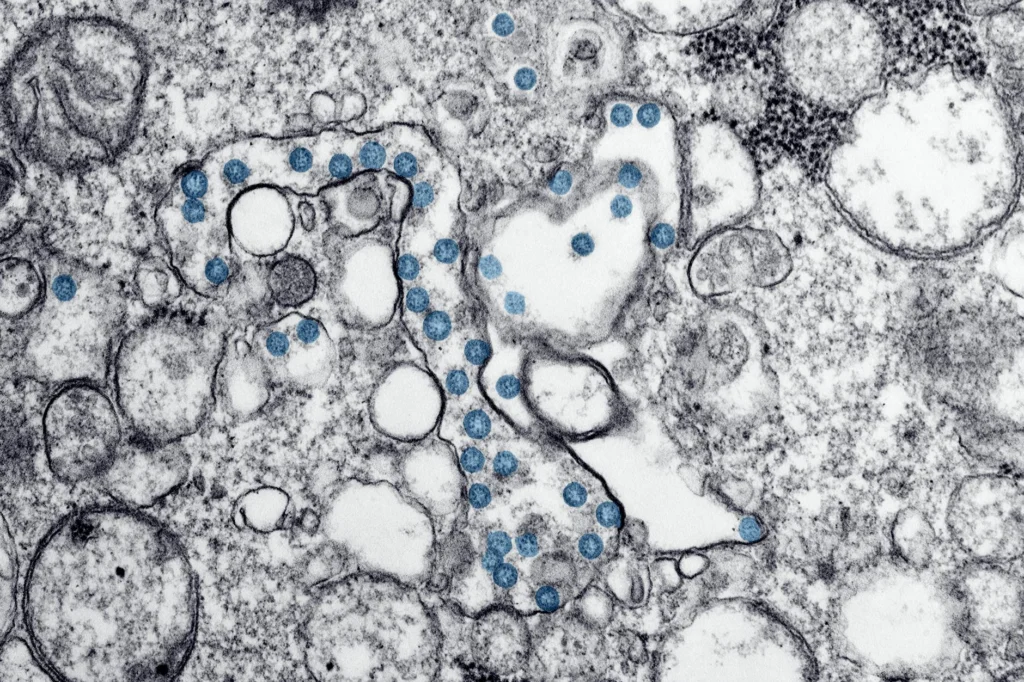
Within minutes of injection, your innate immune system, the older and less specific branch of immunity, detects the vaccine through pattern recognition receptors (PRRs). These receptors, particularly the Toll-like receptor (TLR) family, scan for molecular signatures associated with pathogens: foreign proteins, lipids, or nucleic acids that do not belong in healthy tissue.
This is not a targeted response. The innate immune system does not know what specific pathogen has arrived. It simply recognizes that something foreign is present, and it raises the alarm. Immune cells at the injection site, especially dendritic cells and macrophages, begin engulfing the vaccine components. They chop up the proteins into small fragments called peptides and display them on their surfaces using molecules called MHC (major histocompatibility complex).
Think of it as a “wanted poster” system. Dendritic cells carry these molecular wanted posters from the injection site to the nearest lymph node, where the real training begins.
The Handoff: From Innate to Adaptive Immunity
The lymph node is where the innate and adaptive immune systems meet. Dendritic cells present their MHC-bound peptide fragments to T cells, specialized white blood cells that can recognize incredibly specific molecular shapes. Each T cell carries a unique receptor on its surface, and out of billions of T cells in your body, only a handful will match the specific peptide being presented.
When a match occurs, that T cell activates. It begins dividing rapidly, producing an army of identical cells. Some become “helper” T cells (CD4+), which coordinate the broader immune response. Others become “killer” T cells (CD8+), which can directly destroy cells displaying the foreign protein.
Helper T cells are particularly crucial because they activate B cells, another type of immune cell that produces antibodies. Without helper T cell signals, most B cells cannot mount a robust antibody response. This cooperation between cell types is why adjuvantsA substance added to vaccines to enhance the immune response, improving protection without being the target antigen itself. in vaccines work by enhancing both antigen presentation and co-stimulatory signals that strengthen these cell-to-cell conversations.
The Antibody Factory: B Cells and Germinal CentersA specialized structure within lymph nodes where B cells undergo rapid mutation and selection to produce high-affinity antibodies after vaccination.
Once activated by helper T cells, B cells undergo one of the most remarkable processes in biology. They migrate to specialized structures within the lymph node called germinal centers, where they enter a cycle of rapid mutation and selection that mirrors evolution on fast-forward.
In the germinal center, B cells divide rapidly while introducing random mutations into the genes encoding their antibody receptors. This process, called somatic hypermutationA process in which B cells rapidly mutate their antibody genes, enabling selection of increasingly precise, high-affinity antibodies., is controlled chaos. Most mutations are useless or even harmful. But some produce antibodies that bind their target more tightly than before.
B cells carrying improved antibodies get survival signals from helper T cells. Those with weaker antibodies do not, and they die. Over days and weeks, this Darwinian selection process produces antibodies of increasingly high quality. It is why the antibodies your body makes after a booster shot are typically more effective than those from the first dose: they have been refined through multiple rounds of mutation and selection.
Memory: The Whole Point
The ultimate product of vaccination is not the initial immune response. It is memory. After the vaccine components are cleared and the acute response winds down, two populations of long-lived cells remain.
Memory B cells are sentinels that can persist for years to decades. If they encounter the same pathogen again, they rapidly differentiate into antibody-producing cells, generating a faster and stronger response than the first time. While a primary immune response takes roughly two weeks to produce detectable antibodies, a memory response generates them in just two to four days.
Memory T cells function similarly. After initial activation, the number of pathogen-specific T cells drops sharply but stabilizes at a level 100 to 1,000 times higher than before vaccination, persisting for the rest of a person’s life. These cells are not just more numerous. They are pre-armed, requiring less stimulation to reactivate and capable of mounting a response before the pathogen can establish a foothold.
Long-lived plasma cells add a third layer of protection. These cells, which take up residence primarily in the bone marrow, continuously secrete low levels of antibodies for decades, even without any further exposure to the antigen. They are why a single childhood vaccination against measles can provide lifelong protection.
Why Boosters Work
A booster dose re-engages the memory cells generated by earlier vaccination. Memory B cells re-enter germinal centers and undergo additional rounds of mutation and selection, further increasing antibody affinity with each successive immunization. The result is antibodies that are not only more abundant but also better at neutralizing the pathogen.
Boosters also expand the memory T cell population. Research on mRNA COVID-19 vaccines showed that secondary immunization produced a striking increase in antigen-specific CD8+ T cells, particularly in the lungs, one of the primary sites of respiratory infection.
The mRNA Revolution
Traditional vaccines use weakened or inactivated pathogens, or purified proteins, to trigger an immune response. mRNA vaccines take a fundamentally different approach. Instead of delivering a pre-made antigen, they deliver genetic instructions, wrapped in lipid nanoparticlesMicroscopic fat particles used to package and deliver genetic material into cells. The main vehicle for in vivo CRISPR delivery, with a natural affinity for liver cells. composed of four types of lipids: ionizable lipids, helper lipids, cholesterol, and PEG-conjugated lipids.
Once inside a cell, the mRNA is translated by the cell’s own protein-making machinery into the target antigen (for COVID-19 vaccines, the spike protein). Because the protein is produced inside the cell rather than delivered externally, it gets processed through the same pathway the body uses for virus-infected cells. This means it is displayed on MHC class I molecules, strongly activating killer T cells in addition to the antibody response.
This is a key advantage. mRNA vaccines efficiently induce both cytotoxic T cells and tissue-resident memory T cells through endogenous antigen presentation, something that is harder to achieve with traditional inactivated vaccines.
Beyond Individual Protection: Trained ImmunityThe ability of innate immune cells to respond more effectively to future infections after prior exposure, without relying on classical antibody-based memory.
One of the more surprising discoveries in recent immunology is that some vaccines appear to train the innate immune system as well, not just the adaptive arm. This phenomenon, called trained immunity, involves long-lasting changes to innate immune cells through epigenetic and metabolic reprogramming.
The BCG vaccine (against tuberculosis) is the most studied example. People vaccinated with BCG show enhanced resistance not just to tuberculosis but to unrelated infections as well, suggesting that the vaccine induces a more broadly vigilant innate immune system. This is not traditional immunological memory. It operates through different mechanisms entirely, modifying how genes in innate immune cells are read without changing the DNA sequence itself.
What Can Go Wrong
No system this complex works perfectly every time. Some people generate weaker immune responses due to age, immunosuppression, or genetic variation. The elderly, in particular, tend to produce fewer germinal center reactions and lower-quality antibodies, which is why vaccine formulations for older adults often include stronger adjuvants.
Pathogens can also evolve to escape immune memory. Influenza mutates its surface proteins so rapidly that last year’s antibodies may not recognize this year’s strain. SARS-CoV-2 variants with mutations in the spike protein can partially evade antibodies generated against earlier strains. However, T cells targeting conserved viral regions maintain recognition across variants, which is why vaccinated individuals are still largely protected against severe disease even when antibody effectiveness drops.
This is a critical point often lost in public discussion: antibodies are not the only measure of vaccine protection. T cell immunity provides a durable second line of defense that is harder for pathogens to escape.
This article is for informational purposes only and does not constitute professional medical advice.
Vaccination initiates a coordinated molecular cascade spanning innate pattern recognition, antigen processing and presentation, lymphocyte activation, germinal centerA specialized structure within lymph nodes where B cells undergo rapid mutation and selection to produce high-affinity antibodies after vaccination. reactions, and the establishment of immunological memory. Each stage involves distinct receptor-ligand interactions, signaling pathways, and cellular differentiation programs. What follows is a mechanistic account of how vaccines convert a transient antigen encounter into durable immune protection.
Innate Sensing: Pattern Recognition Receptors and Danger Signals
The immune response to vaccination begins with innate sensing. Toll-like receptors (TLRs), a family of pattern recognition receptors conserved from Caenorhabditis elegans to mammals, detect pathogen-associated molecular patterns (PAMPs) present in or generated by vaccine components. TLRs are type I integral membrane glycoproteins with leucine-rich repeat extracellular domains and cytoplasmic Toll/IL-1R (TIR) homology signaling domains.
Different TLRs recognize distinct molecular classes. TLR1/2/6 heterodimers detect bacterial lipopeptides. TLR4 (with MD-2 and CD14 co-receptors) recognizes lipopolysaccharide. TLR3 detects double-stranded RNA. TLR7/8 sense single-stranded RNA, and TLR9 recognizes unmethylated CpG DNA motifs. Surface TLRs (1, 2, 4, 5, 6) primarily detect microbial membrane components, while endosomal TLRs (3, 7, 8, 9) detect nucleic acids that require internalization for signaling.
TLR signaling proceeds through two major adaptor pathways. The MyD88-dependent pathway (used by all TLRs except TLR3) activates NF-kB and AP-1 transcription factors, driving production of pro-inflammatory cytokinesSmall signaling proteins released by immune cells to coordinate inflammation. Elevated levels are consistently found in patients with depression. (IL-6, TNF-alpha, IL-1beta, IL-12). The TRIF-dependent pathway (TLR3 and TLR4) activates IRF3, inducing type I interferons (IFN-alpha/beta). These two pathways produce different cytokine profiles that shape downstream adaptive immunity: MyD88 signaling via surface TLRs tends to drive Th1 or Th2 polarization, while TRIF-dependent signaling promotes Th1 responses and cross-presentation to CD8+ T cells.
Beyond TLRs, cytoplasmic sensors also contribute. RIG-I and MDA5 detect cytoplasmic RNA, while the cGAS-STING pathway senses cytoplasmic DNA. For mRNA vaccines specifically, the CD8+ T cell response to BNT162b2 was shown to be dependent on type I interferon-dependent MDA5 signaling, not TLR-mediated sensing. The m1-pseudouridineA chemically modified form of uridine, one of RNA's four building blocks. Substituting it into synthetic mRNA prevents the immune system from destroying the mRNA before it can work. modification in BNT162b2 mRNA dampens TLR3/7 and RIG-I recognition while increasing translational efficiency.
AdjuvantsA substance added to vaccines to enhance the immune response, improving protection without being the target antigen itself. as Signal Amplifiers
Adjuvants enhance adaptive immunity by promoting two categories of signals from antigen-presenting cells: signal 1 (antigen peptide-MHC complexes) and signal 2 (co-stimulatory molecules CD40, CD80, CD86 and inflammatory cytokines). Aluminum salts, the oldest licensed adjuvant, promote antigen depot formation and activate inflammatory dendritic cells through uric acid release from necrotic cells. The oil-in-water emulsion MF59, first licensed in Europe in 1997 after seven decades of aluminum salt monopoly, recruits and activates APCs at the injection site. AS03 (alpha-tocopherol + squalene emulsion) activates the IRE1alpha/TRAF2/ASK1/JNK pathway in myeloid cells. AS04 combines aluminum hydroxide with the TLR4 agonist monophosphoryl lipid A (MPL).
mRNA vaccines are somewhat unique in that the mRNA itself is inherently immunostimulatory and is recognized by numerous pattern recognition receptors, allowing mRNA-based vaccines to induce robust responses without requiring additional adjuvants. The lipid nanoparticle carrier also contributes adjuvant activity through activation of inflammasome and NF-kB pathways.
Antigen Processing and MHC Presentation
Activated dendritic cells process vaccine antigens through two major pathways, each loading peptides onto different MHC molecules that activate different T cell populations.
In the exogenous pathway, phagocytosed antigens are degraded in endosomal/lysosomal compartments by cathepsins and other proteases. The resulting peptides (typically 13-25 amino acids) are loaded onto MHC class II molecules and presented to CD4+ helper T cells. This is the dominant pathway for protein subunit and inactivated vaccines.
In the endogenous pathway, proteins synthesized within the cell are degraded by the proteasome. Peptides (typically 8-10 amino acids) are transported into the endoplasmic reticulum by TAP (transporter associated with antigen processing), loaded onto MHC class I molecules, and presented to CD8+ cytotoxic T cells. This pathway is naturally engaged when cells are infected by a virus, and it is why mRNA vaccines efficiently induce multi-epitopeA specific region on a protein molecule that the immune system recognizes as a target. In celiac disease, particular epitopes on wheat proteins trigger an immune attack on the gut lining.-specific cytotoxic T lymphocytes through endogenous antigen presentation: the cell’s own ribosomes translate the mRNA into protein, which enters the proteasomal degradation pathway.
A third mechanism, cross-presentation, allows dendritic cells to load exogenous antigens onto MHC class I, thereby activating CD8+ T cells against threats the dendritic cell has not itself been infected by. Cross-presentation occurs through either a cytosolic pathway (antigen escapes the endosome into the cytosol for proteasomal processing) or a vacuolar pathway (processing occurs within endocytic compartments). This mechanism is critical for non-replicating vaccines that need to generate CD8+ T cell responses.
T Cell Activation: The Three-Signal Model
Naive T cell activation requires three coordinated signals. Signal 1: TCR engagement with peptide-MHC complex, providing antigen specificity. Signal 2: co-stimulatory receptor binding (CD28 on T cells engaging CD80/CD86 on APCs), preventing anergy. Signal 3: cytokine-mediated polarization that directs T cell differentiation.
The cytokine environment determines CD4+ T cell fate. IL-12 and IFN-gamma drive Th1 polarization (cell-mediated immunity, IgG2a isotype switching). IL-4 drives Th2 polarization (humoral immunity, IgE/IgG1 switching). IL-6 and TGF-beta promote Th17 differentiation. IL-6 and IL-21 promote T follicular helper (Tfh) cell differentiation, which is critical for germinal center reactions.
CD8+ T cell activation follows the same three-signal requirement but with MHC class I providing signal 1. Activated CD8+ T cells differentiate into cytotoxic T lymphocytes (CTLs) that kill target cells through perforin/granzyme-mediated apoptosis. BNT162b2 vaccination produced a striking increase in antigen-specific CD8+ T cells after secondary immunization, with a median of 10% MHC-I tetramer-positive CD8+ T cells in the lung at day 42. The CD4+ response was primarily Th1-polarized, with minimal IL-4 induction.
The Germinal Center Reaction: Affinity Maturation at Molecular Resolution
The germinal center (GC) is the structure where B cell responses are refined from broad, low-affinity recognition to precise, high-affinity targeting. B cells within the GC compete for limiting T cell-derived signals, including cytokines and CD40 ligand, that promote their migration between the light zone and dark zone.
The GC has a defined spatial architecture. In the dark zone, B cells (centroblasts) undergo rapid clonal expansion and somatic hypermutationA process in which B cells rapidly mutate their antibody genes, enabling selection of increasingly precise, high-affinity antibodies. (SHM) of their immunoglobulin variable-region genes. Activation-induced cytidine deaminase (AID) introduces point mutations at a rate of approximately 10^-3 per base pair per division, roughly a million-fold higher than the background mutation rate.
Mutated B cells then migrate to the light zone, where they test their new receptors against antigen displayed on follicular dendritic cells (FDCs). B cells with higher-affinity receptors capture and endocytose more antigen, presenting more peptide-MHC II complexes to Tfh cells. This higher peptide-MHC density gives them a competitive advantage for T cell help, mediated through a feed-forward loop of CD40:CD40L signaling.
This competitive selection produces two divergent fates. High-affinity B cells receiving strong Tfh signals downregulate Bcl-6 and upregulate IRF-4/Blimp-1, committing to plasma cell differentiation. Lower-affinity B cells that receive weaker T cell help maintain Bach2 expression and differentiate into memory B cells. This counterintuitive arrangement, where memory B cells tend to emerge from the GC before plasma cells and carry fewer somatic mutations, ensures that the memory compartment retains breadth and flexibility, while plasma cells provide precision.
The GC B cell response can persist in draining lymph nodes for at least six months in some individuals following mRNA vaccination, continuing to produce affinity-matured output cells long after the antigen itself has been cleared.
Immunological Memory: Architecture of Long-term Protection
Durable vaccine protection depends on three distinct memory populations, each operating on different timescales and through different mechanisms.
Long-lived Plasma Cells
Long-lived plasma cells (LLPCs) persist in the bone marrow for decades in the absence of antigen, continuously secreting antibodies into the bloodstream. Their survival depends on niche factors produced by stromal cells, eosinophils, and macrophages in the bone marrow microenvironment. LLPCs provide the first line of humoral defense: pre-existing circulating antibodies that can neutralize a pathogen before it establishes infection.
Memory B Cells
Memory B cells increase in frequency 10 to 100-fold after priming and produce antibodies of higher average affinity than naive B cells. They express higher levels of MHC class II, facilitating antigen uptake and presentation at lower antigen concentrations. Upon re-exposure, memory B cells can either rapidly differentiate into antibody-secreting cells (providing fast humoral protection within two to four days rather than the two weeks required for a primary response) or re-enter germinal centers for additional affinity maturation.
The capacity for GC re-entry is immunologically significant. It means that booster vaccination does not simply expand existing memory but actively improves it, with antibody affinity continuing to increase with each successive immunization as memory B cells undergo further rounds of somatic hypermutation and selection.
Memory T Cells
After the effector phase resolves, antigen-specific T cells persist at 100 to 1,000-fold above pre-immunization frequencies for the remainder of the host’s life. Memory T cells divide into functional subsets: central memory T cells (Tcm, CD62L+CCR7+) recirculate through lymph nodes and rapidly proliferate upon re-encounter; effector memory T cells (Tem, CD62L-CCR7-) patrol peripheral tissues and mount immediate cytokine responses; and tissue-resident memory T cells (Trm, CD69+CD103+) remain stationed in barrier tissues like the lung and gut mucosa.
This layered architecture, pre-formed antibodies from LLPCs, rapid B cell recall, and tissue-stationed T cells, provides overlapping and redundant defense systems that collectively make re-infection far less likely to cause severe disease.
mRNA Vaccines: Engineering the Endogenous Pathway
The ionizable lipid is the critical functional component. At physiological pH (~7.4), it carries a neutral charge, enabling favorable pharmacokinetics. In the acidic endosomal environment (~pH 5-6), it becomes protonated, interacting with the anionic endosomal membrane to promote hexagonal phase formation and cargo release into the cytoplasm.
Once in the cytoplasm, the mRNA is translated by host ribosomes into the encoded antigen. For BNT162b2, the mRNA incorporates N1-methylpseudouridine (m1-psi) modifications that dampen TLR3/7 and RIG-I-mediated inflammatory responses while increasing translational efficiency and biological stability. The expressed spike protein enters the endogenous MHC class I presentation pathway (proteasome, TAP, ER loading) as well as being secreted and taken up by neighboring APCs for MHC class II presentation and cross-presentation.
This dual presentation explains the breadth of the immune response: BNT162b2 induced both robust germinal center B cell and Tfh cell responses and potent CD8+ T cell responses, with the innate immune response to the second dose being markedly stronger than the first, characterized by enhanced IFN-gamma production from natural killer cells and CD8+ T cells in draining lymph nodes.
Trained ImmunityThe ability of innate immune cells to respond more effectively to future infections after prior exposure, without relying on classical antibody-based memory.: Epigenetic Reprogramming of Innate Cells
Classical immunological memory is a property of the adaptive immune system. However, a growing body of evidence demonstrates that innate immune cells can also develop a form of memory. Trained immunity involves long-lasting epigenetic and metabolic reprogramming of innate immune cells such as monocytes and macrophages, resulting in enhanced responsiveness to subsequent, unrelated infections.
The BCG vaccine is the canonical example. Vaccination induces changes in histone modifications (particularly H3K4me3 and H3K27ac marks at promoters of inflammatory genes) and shifts in cellular metabolism (increased glycolysis, altered mevalonate pathway activity) that persist for months to years. These changes are propagated through hematopoietic progenitor cells in the bone marrow, providing a mechanism for their longevity despite the relatively short lifespan of individual monocytes.
Trained immunity is mechanistically distinct from adaptive memory: it lacks antigen specificity, does not involve clonal selection or receptor rearrangement, and operates through chromatin remodeling rather than gene recombination. Its contribution to vaccine-mediated protection, particularly the heterologous effects observed with BCG and other live vaccines, represents an area of active investigation with potential implications for vaccine design.
Variant Escape and the Durability Problem
Antibody-mediated immunity is vulnerable to antigenic drift. When a pathogen mutates its surface proteins, pre-existing antibodies may fail to bind. This is why influenza vaccines require annual reformulation and why SARS-CoV-2 variants with spike protein mutations showed reduced neutralization by vaccine-elicited sera.
T cell immunity, however, is more resilient. Whole-genome T cell epitope scanning shows that approximately 60% of CD8+ T cell responses target non-structural proteins such as ORF1ab, regions that are conserved in more than 85% of the Omicron variant. Because T cells recognize intracellular protein fragments (processed through the proteasome) rather than surface conformations, they survey a much broader set of viral proteins, many of which are under stronger functional constraints and mutate less readily.
This bifurcated vulnerability, where antibodies are strain-specific but T cells are broadly cross-reactive, explains a consistent epidemiological observation: vaccinated individuals may become infected by new variants, but they are substantially less likely to develop severe disease. The antibody wall may be breached, but the cellular defense holds.
This article is for informational purposes only and does not constitute professional medical advice.