Alzheimer’s disease research has been going on for over a century. In that time, the disease has become the most common form of dementia, which now affects 57 million people worldwide and costs the global economy over $1.3 trillion a year. Yet after hundreds of clinical trials, we still have no drug that can stop or reverse it. This is the story of how we got here, and why progress has been so painfully slow.
Alzheimer’s Disease Research Began With a Lecture Nobody Cared About
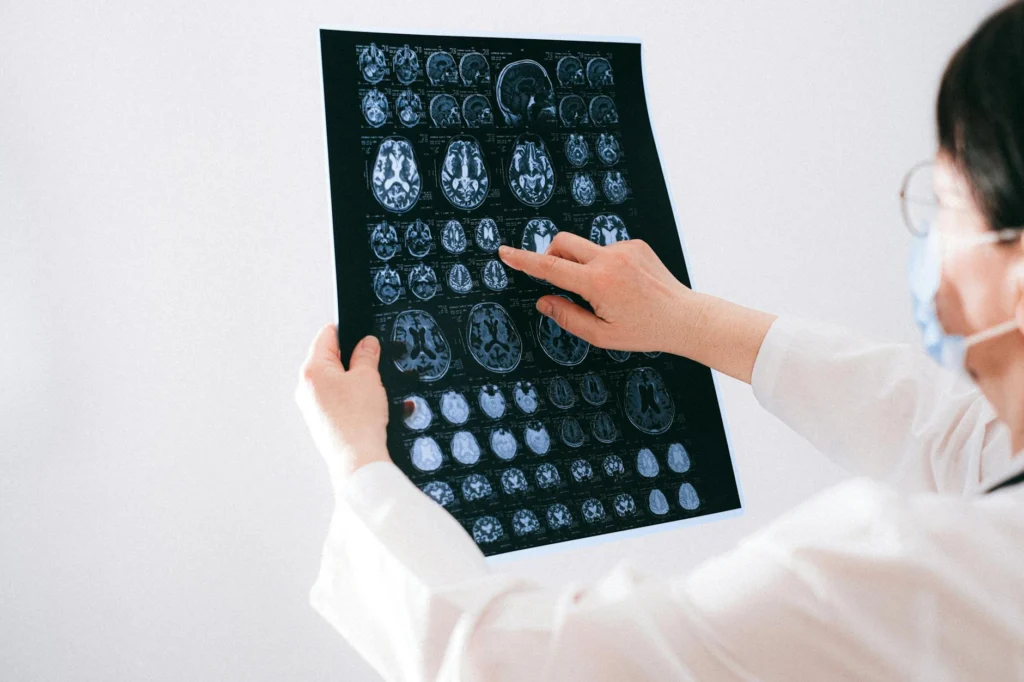
On November 3, 1906, a German psychiatrist named Alois Alzheimer stood before his colleagues at the 37th Meeting of South-West German Psychiatrists in Tubingen. He described the case of Auguste Deter, a woman he had followed from age 50 until her death five years later. She had suffered from paranoia, progressive memory loss, aggression, confusion, and disturbed sleep. When Alzheimer examined her brain after death, he found something striking: distinctive plaques and neurofibrillary tanglesTwisted protein fibers found inside brain cells of Alzheimer's patients, one of the disease's hallmark features. scattered through the tissue.
The audience showed no interest. The chairman moved on without asking for questions or comments. The public press devoted two lines to the lecture while dedicating extensive coverage to the psychoanalytical talks that followed. It was not until 1910 that Emil Kraepelin, Alzheimer’s mentor, included the condition in his influential psychiatry textbook and gave it the name we know today.
Alzheimer himself died in 1915, aged 51, long before his name became a household word.
The Theory That Took Over Everything
For decades after Alzheimer’s discovery, the plaques and tangles he found were known but poorly understood. That changed in the early 1990s. In 1992, researchers John Hardy and Gerald Higgins proposed what became known as the amyloid cascade hypothesisThe theory that amyloid-beta protein buildup in the brain triggers a chain reaction leading to neuronal death and Alzheimer's disease.: the idea that a protein fragment called amyloid-betaA protein fragment that forms sticky clumps in the brains of Alzheimer's patients, believed to be toxic to neurons. builds up in the brain, forms toxic clumps, and triggers a chain reaction of damage that ultimately kills neurons and causes dementia.
The hypothesis was simple, elegant, and convincing. It gave the field a clear target. Pharmaceutical companies poured billions into developing drugs that would remove amyloid from the brain, break up amyloid plaques, or stop amyloid production in the first place. For the next three decades, the amyloid hypothesis dominated Alzheimer’s disease research, shaping what got funded, what got published, and what got ignored.
The problem was that almost none of the drugs worked.
A 99.6% Failure Rate
Between 2002 and 2012, 413 Alzheimer’s clinical trials were conducted, testing 244 different compounds across Phase 1, 2, and 3. The overall success rate was 0.4%. That means 99.6% of all tested drugs failed, one of the highest failure rates in any disease area. For context, the failure rate for cancer drugs during the same period was around 81%.
The pattern continued after 2012. A comprehensive review covering 2004 through 2021 found 98 unique compounds that failed in Phase 2 and 3 trials during that period. Only two received any form of approval, giving a success rate of roughly 2%. Drug after drug cleared amyloid from the brain exactly as designed, yet patients kept declining at roughly the same rate as those on placebo.
One major contributing factor: up to 50% of patients with mild cognitive impairmentNoticeable cognitive decline beyond normal aging that does not severely interfere with daily functioning. enrolled in these trials may not have had amyloid abnormalities at all. Many trials were testing amyloid-clearing drugs on people who might not have had an amyloid problem in the first place.
A Landmark Paper Built on Doctored Images
In 2006, a team led by Sylvain Lesne published a paper in Nature claiming to have identified a specific amyloid protein, called Aβ*56, that directly caused memory impairment in mice. The paper was hugely influential, cited nearly 2,500 times. It gave the amyloid hypothesis a specific, actionable target and helped direct millions in research funding.
In 2022, a Science investigation revealed evidence that the paper’s key Western blot images had been digitally manipulated. In 2024, nearly all the authors agreed to retract the paper. The senior author, Karen Ashe, acknowledged the manipulations, though she maintained the conclusions were still valid. Lesne, the first author, was the sole holdout against retraction. He resigned from the University of Minnesota in early 2025 after an institutional investigation flagged data integrity concerns in multiple papers.
Some scientists argued the fraud supported broader doubts about the amyloid hypothesis. Others insisted the hypothesis stood on far more evidence than one paper. Either way, it was a credibility blow to a field already struggling with public trust.
The New Drugs: Modest Gains, Real Risks
After decades of failure, two anti-amyloid antibodies finally showed measurable results. Lecanemab received full FDA approval in July 2023, and donanemab followed in July 2024. Both work by binding to and clearing amyloid from the brain.
A meta-analysis of clinical trial data confirmed that both drugs outperformed placebo on cognitive and functional measures. Lecanemab slowed cognitive decline by about 27% over 18 months. Donanemab showed about a 35% lower risk of disease progression.
But these numbers require context. A 27% slowing of decline does not mean patients improved or even stabilized. It means they got worse slightly more slowly than the placebo group. The clinical difference on standard cognitive tests was small enough that some researchers question whether patients or their families would notice it in daily life.
The safety profile raised additional concerns. The risk of amyloid-related imaging abnormalities (ARIAAmyloid-related imaging abnormalities, including brain swelling and bleeding, that can occur as side effects of Alzheimer's drugs.), which include brain swelling and microbleeds, was 4.35 times higher in treated patients than in the placebo group. About one in five patients on lecanemab developed these abnormalities. Some cases proved fatal.
Meanwhile, aducanumab, which was controversially approved by the FDA in 2021 against the recommendation of its own advisory committee, was withdrawn from the US market in February 2024 due to costs and limited therapeutic benefit.
Why One Hypothesis Is Not Enough
The fixation on amyloid has always had skeptics. Cognitively normal elderly people can have brains full of amyloid plaques. Some Alzheimer’s patients have relatively little amyloid. The correlation between plaque load and cognitive decline is weak.
Today, at least seven major hypotheses compete to explain what drives the disease: the amyloid cascade, tau hyperphosphorylation, neuroinflammationInflammation within the brain or nervous system, driven by immune cells called microglia. Increasingly linked to depression, anxiety, and other psychiatric disorders., oxidative stress, mitochondrial dysfunction, cholinergic dysfunction, and vascular dysfunction. None of them are mutually exclusive. Many researchers now believe Alzheimer’s is not one disease but a constellation of overlapping processes that converge on the same outcome: neurons dying faster than they should.
Even John Hardy, who co-authored the original amyloid cascade hypothesis in 1992, acknowledged in 2022: “When we found amyloid mutations I thought, and the field thought, that sorting out amyloid was to sort out dementia. We had this idea of a magic bullet. We do not think that anymore.”
The Scale of What Is Coming
While the scientific debate continues, the demographic clock does not wait. The WHO reports nearly 10 million new dementia cases every year, and Alzheimer’s accounts for 60 to 70% of them. Projections suggest the number of people with dementia will reach 78 million by 2030 and 139 million by 2050, with costs projected to reach $2.8 trillion annually by 2030.
In the United States alone, Alzheimer’s-related mortality increased by 141% between 2001 and 2021. During the same period, deaths from heart disease, the leading killer, fell by 2.1%. We are getting better at keeping people alive long enough to develop the one disease we still cannot meaningfully treat.
The honest assessment is uncomfortable. We are over a century into Alzheimer’s disease research and still arguing about whether we have identified the right mechanism. The two approved disease-modifying drugs offer a modest slowing of decline at the cost of significant side effects. The pipeline has 138 drugs in clinical trials, but the historical failure rate suggests most of them will not make it.
What the field needs is not another round of the same bet. It needs the intellectual humility to accept that a disease affecting the most complex organ in the human body probably does not have a single cause, a single mechanism, or a single solution.
More than a century of Alzheimer’s disease research has produced a remarkably specific kind of failure: not a failure of effort, funding, or scientific talent, but a failure of paradigm. With 57 million people living with dementia globally and 10 million new cases each year, the gap between the scale of the problem and our mechanistic understanding of it remains vast.
From Histopathology to Hypothesis: Alzheimer’s Disease Research Through the Decades
Alois Alzheimer’s 1906 presentation to the 37th Meeting of South-West German Psychiatrists described the case of Auguste Deter: progressive cognitive decline, paranoia, sleep disturbance, and aggressive behavior from age 50 until her death at 55. Post-mortem histological examination using the then-novel Bielschowsky silver stain revealed distinctive extracellular plaques and intracellular neurofibrillary tanglesTwisted protein fibers found inside brain cells of Alzheimer's patients, one of the disease's hallmark features. in the cerebral cortex. The finding generated essentially no interest from the scientific audience. It took Kraepelin’s inclusion of “Alzheimer’s disease” in his 1910 Psychiatrie textbook to establish the eponymous diagnosis.
For the better part of the 20th century, AD was considered a rare presenile dementia, distinct from “senility.” The reconceptualization of AD as the primary cause of age-related dementia came gradually through the 1970s and 1980s, driven by epidemiological data and the biochemical characterization of plaque and tangle components.
The Amyloid Cascade HypothesisThe theory that amyloid-beta protein buildup in the brain triggers a chain reaction leading to neuronal death and Alzheimer's disease. and Its Molecular Foundations
The identification of amyloid-betaA protein fragment that forms sticky clumps in the brains of Alzheimer's patients, believed to be toxic to neurons. (Aβ) as the primary constituent of senile plaques in the mid-1980s, followed by the mapping of the amyloid precursor protein (APP) gene to chromosome 21, created a compelling genetic narrative. Individuals with trisomy 21 (Down syndrome) carry three copies of APP and frequently develop AD neuropathology by age 40. Familial early-onset AD mutations were subsequently found in APP itself and in presenilin-1 (PSEN1) and presenilin-2 (PSEN2), the catalytic subunits of gamma-secretase that cleaves APP to produce Aβ.
In 1992, Hardy and Higgins formalized these observations into the amyloid cascade hypothesis, proposing that Aβ accumulation is the initiating event in AD pathogenesis, triggering tau hyperphosphorylation, neuroinflammationInflammation within the brain or nervous system, driven by immune cells called microglia. Increasingly linked to depression, anxiety, and other psychiatric disorders., synaptic loss, and neuronal death in a linear cascade. The hypothesis was refined through subsequent reappraisals in 2002, 2006, 2009, and 2016, with a shift from insoluble fibrils to soluble Aβ oligomers as the putative toxic species. The core claim, that amyloid is upstream of all other pathology, remained intact.
The hypothesis had extraordinary consequences for resource allocation. Anti-Aβ therapies dominated the clinical pipeline for three decades. Of 146 disease-modifying compounds tested in clinical trials from 2002 to 2012, 70 targeted amyloid-beta, compared with 13 targeting tau-related mechanisms and 62 pursuing neuroprotective strategies.
The Trial Failure Landscape: 99.6% Attrition
A systematic analysis of Clinicaltrials.gov data from 2002 to 2012 documented 413 AD trials testing 244 unique compounds, with an overall success rate of 0.4%. The 99.6% failure rate represented the highest attrition of any major therapeutic area, exceeding cancer (81%) by a wide margin.
An exhaustive review covering 2004 through 2021 identified 98 unique phase II and III compounds that failed, against two that received any regulatory approval, yielding a 2.0% success rate. The failures were not exclusively amyloid-targeting drugs, but the amyloid-centric pipeline bore the brunt.
The methodological post-mortems identified several systemic problems. Up to 50% of MCI patients and 20% of mild AD patients enrolled in trials lacked amyloid pathology on PET imaging, meaning they may not have had AD at all. Small molecule candidates often lacked demonstrated blood-brain barrierA selective membrane that controls what substances can pass from the bloodstream into the brain. Nanoplastics are small enough to cross this barrier, allowing them to accumulate in brain tissue. penetration. Many pivotal trials were initiated without sufficient phase II evidence. Preclinical animal models, typically APP/PSEN1 transgenic mice, recapitulate amyloidosis but lack tau pathology, neuronal death, and the full inflammatory milieu characteristic of human AD.
The Aβ*56 Fraud and Its Fallout
In 2006, Lesne et al. published a Nature paper claiming that a specific soluble Aβ assembly, designated Aβ*56, was present in transgenic mouse brains in proportion to cognitive decline and, when injected into rats, induced memory impairment. The paper was cited nearly 2,500 times and directed substantial research funding toward Aβ oligomer biology.
A 2022 Science investigation found evidence of systematic image manipulation in the paper’s Western blots. In 2024, Nature retracted the paper after nearly all authors agreed to the retraction. Senior author Karen Ashe acknowledged the manipulations but maintained that the experimental conclusions remained valid. First author Sylvain Lesne, the sole dissenter, resigned from the University of Minnesota in early 2025 after institutional investigation found data integrity concerns across multiple publications.
The retraction did not invalidate the broader amyloid hypothesis, which rests on genetic, biochemical, and clinical evidence beyond one paper. But it removed a key empirical pillar for the specific oligomer-toxicity model and reinforced concerns about the evidentiary standards in the field.
Lecanemab and Donanemab: What the Data Actually Show
After decades of failure, two anti-Aβ monoclonal antibodies demonstrated statistically significant efficacy. Lecanemab (Leqembi), an IgG1 antibody targeting Aβ soluble protofibrils, received full FDA approval in July 2023. Donanemab, targeting pyroglutamate-modified Aβ in deposited plaques, was approved in July 2024.
A 2025 meta-analysis pooling five clinical studies (n=4,824) confirmed that both drugs outperformed placebo on CDR-SB, ADAS-Cog 14, ADCOMS, and amyloid burden on PET. The CLARITY-AD trial showed lecanemab slowed cognitive decline by approximately 27% over 18 months as measured by CDR-SB. The TRAILBLAZER-ALZ 2 trial reported about 35% slower progression for donanemab.
The clinical meaningfulness of these effect sizesA standardized measure of the magnitude of difference between groups in a study, independent of sample size. remains contested. The absolute CDR-SB difference between lecanemab and placebo at 18 months was approximately 0.45 points on an 18-point scale. Whether this translates to perceptible benefit for patients or caregivers is an open question. Some researchers characterize the results as demonstrating proof of concept for amyloid removal; others argue the modest benefit at substantial risk does not constitute a meaningful disease-modifying therapy.
Safety data added to the debate. The risk of amyloid-related imaging abnormalities (ARIAAmyloid-related imaging abnormalities, including brain swelling and bleeding, that can occur as side effects of Alzheimer's drugs.) was 4.35 times higher in treated groups, with ARIA-E (edema) and ARIA-H (hemorrhage/hemosiderin deposition) both significantly elevated. APOE ε4 carriers faced disproportionately higher risk. Superficial siderosis of the central nervous system was notably increased.
The cautionary tale of aducanumab remains instructive. Approved by the FDA in 2021 against the near-unanimous recommendation of its advisory committee, based on the surrogate endpoint of amyloid plaque reduction rather than clinical benefit, it was withdrawn from the market in February 2024 after the European Medicines Agency rejected it and the costs proved unsustainable.
Beyond Amyloid: The Multifactorial Reality
The fundamental challenge is that late-onset sporadic AD, which accounts for 94-99% of cases, may not be driven by the same mechanism as the rare familial forms that generated the amyloid hypothesis. Several unresolved observations undermine a purely amyloid-centric model: Aβ deposition occurs in cognitively normal elderly individuals; the correlation between plaque burden and cognitive status is weak; and pre-clinical models biased toward the amyloid hypothesis have not predicted clinical success.
Current understanding increasingly favors a multifactorial model incorporating at least seven overlapping pathogenic pathways: the amyloid cascade, tau hyperphosphorylation, neuroinflammation (particularly microglial activation and cytokine release), oxidative stress, mitochondrial dysfunction (impaired bioenergetics and quality control), cholinergic degeneration, and cerebrovascular dysfunction. APOE ε4, the strongest genetic risk factor for late-onset AD, operates across multiple pathways including cholesterol metabolism, Aβ clearance, and neuroinflammatory regulation.
Pure AD, characterized exclusively by amyloid plaques and tau tangles, is rare at autopsy. Most cases exhibit mixed pathologies, including Lewy body deposits, TDP-43 inclusions, and vascular lesions. This heterogeneity suggests that what we call Alzheimer’s disease may encompass several distinct or overlapping disease processes converging on a common clinical phenotype.
Even Hardy, who co-authored the 1992 hypothesis, stated in 2022: “When we found amyloid mutations I thought, and the field thought, that sorting out amyloid was to sort out dementia. We had this idea of a magic bullet. We do not think that anymore.”
The Demographic Pressure
The WHO identifies dementia as the seventh leading cause of death globally. Projections estimate 78 million people with dementia by 2030 and 139 million by 2050, with annual costs projected to reach $2.8 trillion by 2030. In the United States, AD-related mortality rose 141% between 2001 and 2021 while heart disease mortality fell 2.1%. We are extending lifespans into the age range where AD incidence doubles every five years past 65, without a corresponding advance in treatment.
The current pipeline contains approximately 138 drugs in 182 clinical trials. Historical attrition rates suggest most will fail. The field needs not just new molecules but a fundamental reorientation: from linear cause-and-effect models toward systems biology approaches that can accommodate the multimodal, heterogeneous reality of neurodegeneration in the aging brain.
After more than a century of Alzheimer’s disease research, the most honest summary remains the one offered by Morgan et al. in 2022: “We simply do not understand the disease well enough.”